Main Text
1 Introduction
The thymus, as a primary central immune organ in the body, plays an essential role in the differentiation, development, and maturation of T cells, and within the immune system [1]. Thymic epithelial cells (TECs) provide a unique microenvironment for various stages of T cell development [2]. The thymus reaches its maximum weight during puberty and then undergoes a process of atrophy, also being one of the earliest organs to exhibit signs of aging [3]. These changes ultimately lead to a decline in the function of thymus-nurtured T lymphocytes, restricted replication of memory T lymphocytes, obstruction of the peripheral T lymphocyte recirculation to the thymus pathway, and an apparent reduction in the output capacity of T cells, resulting in the aging of the immune system and impaired responsiveness [4-7]. Therefore, delaying thymic aging is crucial for enhancing overall immunity and delaying the aging process.
Rapamycin (RAPA) is a recognized anti-aging drug [8], with its target, the mechanistic target of rapamycin (mTOR), being a notable regulator of cell growth and metabolism [9]. In mouse models, RAPA has been confirmed to have immunomodulatory functions, primarily by inhibiting the mTOR1 signaling pathway [10]. And mTOR signaling transduction can regulate the activation and differentiation of T cells [11], enhance the immune memory of CD8+T cells, and mediate CD4+ regulatory and non-regulatory T cells, which, however, may also weaken the body's defense against acute viral and bacterial infections [9,12]. Thus, exploring how to reasonably utilize RAPA to achieve its beneficial effects is of particular importance. Studies have also found that RAPA, in combination with other anti-aging drugs such as metformin (MET), may effectively extend the lifespan of mice [13]. Additionally, a ketogenic diet has been proven to extend the lifespan of mice by reducing mTORC1 activity [14].
The visceral manifestations theory in traditional Chinese medicine (TCM) proposed “the kidney stores essences, governing yin and yang in the body”. It considers essences in the kidney as the basis of life, which play a decisive role in growth, development, mature and aging of a person. Accordingly, as stated in the ancient book The Spiritual Pivot-Tian Nian, people with longevity have unblocked qi, blood, and meridians, as well as excessive kidney-qi. The modern medical research also indicated that the main immune cells in the human body originate from hematopoietic stem cells in the bone marrow, revealing a close association between the kidneys and immunity. Besides, the thymus and bone marrow, as central immune organs, play a crucial role in regulating human immune function [15]. Multiple studies proved that kidney deficiency triggers atrophy of thymus [16] and influences the development of T cells. Tonifying kidneys is the core principle in anti-aging treatment using TCM. To explore the impact of kidney-tonifying Chinese medicines on thymus, we selected Cynomorium songaricum (CS) [17], Cistanche deserticola (CD) [18], Epimedium brevicornu (EB) [19], Morindae officinalis (MO) [20], and Drynaria fortunei (DF) [21], five representative kidney-tonifying Chinese medicines. We aim to provide more feasible strategies for promoting thymus regeneration and more experimental basis for kidney-tonifying and anti-aging TCM theory by investigating the role of kidney-tonifying Chinese medicine in the acute thymic degeneration and regeneration model induced by RAPA.
2 Materials and methods
2.1 Experimental animals
Male BALB/c mice aged 6-8 weeks were purchased from Slack Company. The mice were reared in a specific pathogen-free (SPF) room with a temperature of 23 ± 1.1 ℃ and a relative humidity of 60 ± 10%. The animal experiments were approved by the Animal Ethics and Welfare Committee of Zhejiang Chinese Medical University (Ethical Approval Number: IACUC-20240311-12) and conducted in accordance with the guidelines for the care and use of laboratory animals (accredited by the Association for Assessment and Accreditation of Laboratory Animal Care International (AAALAC)). After administration, all of the mice were inhaled with 1-2% isoflurane (Veterinary Medicine Standard 031217015, RWD, China) in the RWD Small Animal Anesthesia Machine (R510-11, RWD, China), and subsequently sacrificed by cervical dislocation.
2.2 Experimental medicines
TCM decoction pieces were purchased from the Binjiang Branch of Zhejiang Famous TCM Museum, and were ground into fine powder using a Chinese medicine grinder and sifted through a No. 6 sieve. The samples were then completely immersed in 75% food-grade ethanol for 12 h to ensure thorough penetration. Then, a heating rotary evaporation device was used to perform rotary evaporation, which involved high-temperature heating and rotary evaporation to remove as much ethanol residue from the samples as possible, aiming to achieve a solution suitable for freeze-drying. During the rotary evaporation process, temperature and vacuum were controlled to ensure efficient removal of ethanol while avoiding thermal damage to the samples. Subsequently, the obtained solution was subjected to freeze-drying, during which dehydration was completed under low-temperature and vacuum conditions to dry the samples, with Vacuum freeze dryer (SZFD-50A, Shun Zhi Instrument Manufacturing, Shanghai, China). Finally, the dried samples were sealed and stored at 4 ℃ in a refrigerated environment to maintain their stability and activity. Before use, according to the minimum recommended dosage of CS in “Chinese Pharmacopoeia (2020 edition)” as 5 g (0.65 g/kg), CD as 6 g (0.78 g/kg), EB as 6 g (0.78 g/kg) , MO as 6 g (0.39 g/kg), and DF as 6 g (0.39 g/kg) [22], the medicine was prepared with drinking water for gavage administration. RAPA was procured from Aladdin Biochemical Technology (A2221042, Shanghai, China). 1,1-Dimethylbiguanide hydrochloride (MET) was ordered from Shanghai Bide Pharmaceutical Technology (BSC188, Shanghai, China).
2.3 Method and cycle of medication
A total of 42 healthy male BALB/c mice aged 6-8 weeks were adaptively reared for 3 days, and subjected to depilation on dorsal region of mice using hair removal cream before experiments. Mice were randomly divided into 7 groups (6 mice/group), normal group, RAPA group, natural recovery (N-R) group, MET group, CS group, CD group, EB group, MO group, and DF group. Mice in the normal group received 0.1 mL/10 g normal saline intraperitoneally from day 1 to 5, and 0.1 mL/10 g of drinking water by gavage from day 6 to 10. Mice in other groups were administered with 1 mg/kg RAPA intraperitoneally daily for 5 consecutive days, and were fasted for 12 h on day 5 after the administration. Mice were given 0.1 mL/10 g of drinking water by gavage in N-R group, received 300 mg/kg MET intraperitoneally in MET group, and were administered with extracts of CS, CD and EB by gavage once daily for 5 consecutive days in CS group, CD group and EB group. Mouse weight and food intake were recorded daily.
2.4 Body weight and thymus index
The thymus at the upper part of the anterior mediastinum behind the sternum was obtained. The connective tissue around the thymus was carefully removed and the wet weight of the thymus was weighed using an electronic balance. Thymus index (%) = thymic wet weight (g)/mouse body weight (g) × 100%, and then statistics were performed.
2.5 Flow cytometry
Mice thymus were minced on a plate containing phosphate buffer saline (PBS) (20240805,Solaibao, Beijing, China), and then filtered through a 70 μm cell strainer to create a single-cell suspension. Besides, peripheral blood mononuclear cells (PBMCs) were obtained from peripheral blood using red blood cell lysis solution (3702, Beyotime, Shanghai, China). Different flow cytometry antibodies (0.5 μL/each, BD Pharmingen, San Diego, CA, USA), APC Hamster Anti-Mouse CD3e(2306072), FITC Rat Anti-Mouse CD4 (3095781), PE Rat Anti-Mouse CD8a (5223517), and PerCP-Cy5.5 Hamster Anti-Mouse TCR β (2040773) were added to the thymocyte suspensions and PBMC samples from each group, incubated at 4 ℃ in the dark for 30 min, washed, resuspended in 400 μL PBS, and then filtered through a 200-mesh sieve. Flow cytometer (CytoFLEX Beckman, USA) was used to detect and collect data. Data analysis was performed using CytExpert 2.4 (CytoFLEX Beckman, USA).
2.6 Hematoxylin-eosin (H&E) staining
Thymus were fixed in 10% formalin for 48 h and embedded in paraffin. Tissue sections of 4 μm thickness by LEI-CARM2245 136 microtome (Leica Microsystems Nussloch GmbH, Germany) were prepared for histopathological analysis. Sections were stained with hematoxylin for 1.5 min and eosin for 40 s (20231218, Jiancheng Technology, Nanjing, China). Stained sections were examined and visualized using a Zeiss fluorescence microscope (AXIO SCOPE. A1, Carl Zeiss AG, Germany).
2.7. Immunofluorescence
Paraffin sections of the thymus were subjected to routine dewaxing and hydration procedures. The slides were subsequently immersed in EDTA antigen retrieval solution (50X) (ZLI-9069, ZSGB-Bio, China) and deionized water (1:50 dilution) and deionized water (1:50 dilution), followed by heating in a microwave oven for optimal antigen retrieval. Thereafter, rabbit anti-mouse cytokeratin 8 (CK8: ab53280, Abcam, UK, 1:200 dilution) and mouse monoclonal cytokeratin 5 (CK5: BF0493, Affinity Biosciences, China; 1:200 dilution) were applied and incubated at 4 ℃ for 16 hours. These antibodies were detected using a four-color mIHC fluorescence kit (Recordbio Biological Technology, Shanghai, China) based on tyramide signal amplification (TSA) technology. Co-staining of A and B was performed with this kit. Cell nuclei were labeled with DAPI (P0131, Beyotime, Shanghai, China). Images were captured using a Zeiss upright fluorescence microscope (AXIO SCOPE.A1).
2.8 T-cell receptor excision circles (TRECs) quantification
Peripheral blood genomic DNA was extracted using a mammalian genomic DNA extraction kit (Beyotime, Shanghai, China). TRECs in peripheral blood monocytes were quantified via Real-Time PCR to evaluate thymus vitality. The primers used for the determination were synthesized by Sangon Biotech (Shanghai, China), including TREC, 5′-CATTGCCTTTGAACCAAGCTG-3′ and 5′-TTATGCACAGGGTGCAGGTG-3′.
2.9 RNA isolation, reverse transcription, and quantitative real-time polymerase chain reaction (qRT-PCR)
Thymic RNA was extracted using an RNA extraction kit (RN002PLUS, Shanghai Yishan Biotechnology Co., Ltd., China), and underwent reverse transcription. The mRNA expression was detected using the SYBR Green PCR method. Gene relative quantification was carried out using the 2−ΔΔCt method [23], with GAPDH as the endogenous control. QRT-PCR was conducted with 2×SG FastqPCRMix (B639271; Sangon Biotech,Shanghai ,China). All reagents and primers were sourced from Sangon Biotech Co., Ltd. (Shanghai, China), and the primers used were listed in the last (Table S2).
2.10 Statistical analysis
Unless otherwise specified, all results are presented as the mean ± SEM. All statistical analyses were performed with unpaired Student's t-tests, one-way ANOVA with Tukey's post hoc test, or two-way ANOVA with Tukey's post hoc test using Prism 8.0.2 (GraphPad Software, China). p value of less than 0.05 was considered statistically significant.
3 Results
3.1 Kidney-tonifying Chinese medicines improved health condition of mice
Through comparing the efficacy of different kidney-tonifying Chinese medicines and observing thymus tissue structure, we suggested that MO and DF show no significant changes in the density of cortical thymocytes and the medullary region, while CS, CD, and EB exhibit greater potential for thymus regeneration. So CS, CD, and EB were selected for subsequent analyses (Figure 1A and Table S1). The results indicated that 1mg/kg RAPA induced severely atrophied thymus, manifested as reduced volume and weight of thymus and significantly decreased thymus index. The thymus volume weight and index were increased in the N-R group, MET group, and TCM groups, with the MET group and each TCM groups showing a more evident increase, but no significant difference between N-R group and each treatment group (Figure1 B, C, and D and Table 1). On the 7th day of the experiment, the body weight of mice in the N-R group, MET group, and TCM groups were all increased notably, with the CD groups showing a particularly significant increase. At the same time, the grip strength ability of mice in the TCM groups were elevated, but there was no significant difference between these groups. No remarkable difference in food intake was detected between the groups (Figure1 E, F, and G).
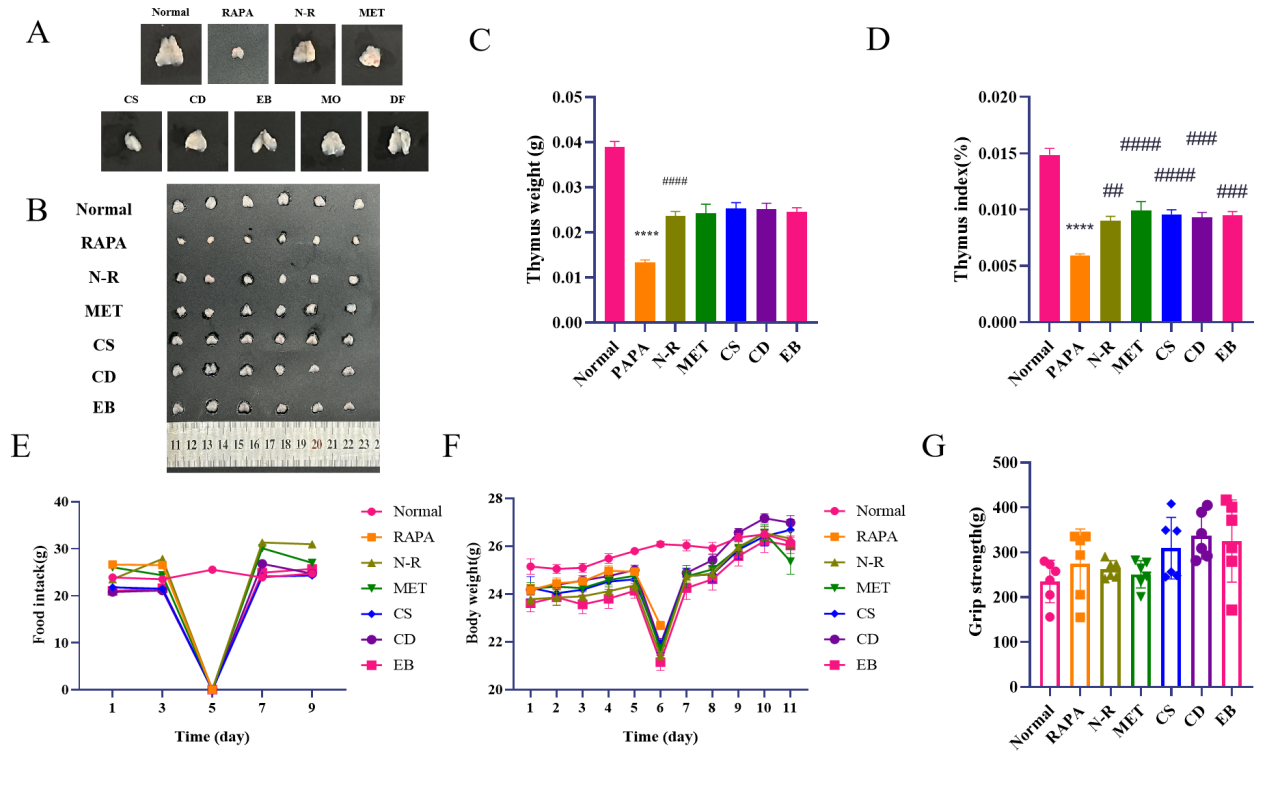
Figure 1 Kidney-tonifying Chinese medicines improved health condition of mice. (A) Representative images of thymus from mice in each group. Group abbreviations: Rapamycin (RAPA), Normal recovery (N-R), Metformin (MET), Cynomorium songaricum (CS), Cistanche deserticola (CD), Epimedium brevicornu (EB), Morindae officinalis (MO), and Drynaria fortunei (DF). (B) Images of thymus from mice in each group. (C) Bar graph of thymus weight in each group. (D) Bar graph of thymic index (thymus weight/mouse body weight × 100) in each group. (E) Bar graph of food intake for mice in each group. (F) Bar graph of body weight for mice in each group. (G) Bar graph of grip strength for mice in each group. Compared to the normal group, *p < 0.05, **p < 0.01, ***p < 0.001, ****p < 0.0001, compared to RAPA group, #p < 0.05, ##p < 0.01, ###p < 0.001, ####p < 0.0001
Table 1 Wet weight, body weight and thymus index of mice.
| Thymus weight (g) | Body weight (g) | Thymus index (%) | |
|---|---|---|---|
| Normal | 0.039 ± 0.003 | 26.2 ± 0.611 | 0.149 ± 0.014 |
| RAPA | 0.013 ± 0.001 | 22.7 ± 0.436 | 0.059 ± 0.004 |
| N-R | 0.024 ± 0.003 | 0.024 ± 0.003 | 0.090 ± 0.010 |
| MET | 0.024 ± 0.005 | 25.4 ± 1.308 | 0.099 ± 0.020 |
| CS | 0.025 ± 0.003 | 26.7 ± 1.067 | 0.096 ± 0.010 |
| CD | 0.025 ± 0.003 | 27.0 ± 0.717 | 0.093 ± 0.010 |
| EB | 0.025 ± 0.002 | 26.0 ± 0.848 | 0.095 ± 0.008 |
3.2 Kidney-tonifying Chinese medicines improved thymus structure of mice
Through H&E staining, the cortex and medulla of the thymus were observed. The data revealed that, compared to the normal group, the RAPA group exhibited a significantly reduced area of the thymic medulla, a disordered and unclear junction between the cortex and medulla, and a substantial loss of cells within the thymus. In the N-R group and all treatment groups, the structures of the thymic cortex and medulla were partially restored, with an increased cell density in the thymic cortex. More epithelial cells were observed in the junction area between the cortex and medulla, clarifying the boundary between these two regions. The MET and CS groups demonstrated a more pronounced increase in the number of thymic cells in the cortex, while the CD and EB groups showed an increase in the medulla, with restored structure and a higher number of medullary epithelial cells (Figure 2).

Figure 2 Kidney-tonifying Chinese medicines improved thymus structure of mice. H&E staining map of thymus in each group of mice: 4 × (top), scale bar=250 μm. Compared to 20 × (bottom), scale bar=50 μm.
3.3 Kidney-tonifying Chinese medicines improved epithelial cell structure and distribution in mice
CK5 and CK8 belong to the CK family, and their expressions are mainly associated with the proliferation, differentiation, and functional activity of thymic epithelial cells (TECs) in thymic tissue [22]. Additionally, CK8 and CK5 serve as specific marker proteins for TECs in the thymic cortex and medulla regions, respectively. Therefore, we performed immunofluorescence staining for CK5 and CK8 in mouse thymus.
The experimental results showed that in the RAPA group, relative to the normal group, the reticular structure of the thymus medulla was significantly altered, with atrophy and a marked loosening of the structure, reduced cell-to-cell connections, and decreased positive expressions of CK5. Compared to the RAPA group, the reticular structure of the medulla in the thymus of the N-R group, MET group, and TCM group was restored. However, the repair of the cortical reticular structure in the N-R group was still incomplete, with reduced fluorescence intensity of CK8 in the cortex. By contrast, the MET group and TCM group showed an increased proportion of the thymic medulla, an elevated number of mTECs, upregulated positive expressions of CK5 and CK8, and a dense reticular structure of cTECs. The thymic capsule, cortex, and deep cortex areas in the MET group, CS group, and CD group presented a significant increase in CK8 fluorescence intensity. In contrast, the EB group exhibited a more pronounced increase in CK5 fluorescence intensity, with clearer reticular nodes of mTECs (Figure 3).

Figure 3 Kidney-tonifying Chinese medicines improved epithelial cell structure and distribution in mice.Spatial distribution of thymic TECs in each group (100×; CK8: green; CK5: red; DAPI: blue). (200×; CK8: green; CK5: red; DAPI: blue). Scale bar=100 μm (top) and 50 μm (bottom).
3.4 Kidney-tonifying Chinese medicine impacted the differentiation of thymic T cell subsets in mice
CD4 and CD8 are markers for T cell subsets, and mature T cells have specific T cell receptors (TCR) and surface markers such as CD3. The differentiation and maturation of thymocytes involve four stages: double-negative (DN) cells (CD4-CD8-), double-positive (DP) cells (CD4+CD8+), single-positive cells, and mature cells (CD4+ (CD4SP) cells (CD3+TCR β+CD4+CD8-) and CD8+ (CD8SP) cells (CD3+TCR β+CD4-CD8+)) [23,24].
To assess the changes in the differentiation of T cell subsets in mouse thymus, flow cytometry analysis of the thymus was conducted. The results proved that in contrast to the normal group, the PAPA group had dramatically increased DN cells, CD3+TCR β+ cells and CD4SP cell subsets, as well as starkly decreased DP and CD8SP cell subsets. In the N-R group, the DN cells, CD3+TCR β+ cells, and CD4SP cell subsets were remarkably reduced, while the DP and CD8SP cell subsets were greatly augmented. Compared with the N-R group, the proportion of DP cell subsets in CS and EB groups increased more in each TCM group. The CD3+TCR β+ cell subset was prominently reduced in all TCM groups, with a more pronounced decrease in the CS and CD groups. The MET group showed an increase in the CD4SP cell subset, and all TCM groups displayed an obvious increase in the CD4SP cell subset and a signally decrease in the CD8SP cell subset (Figure 4 A and B).
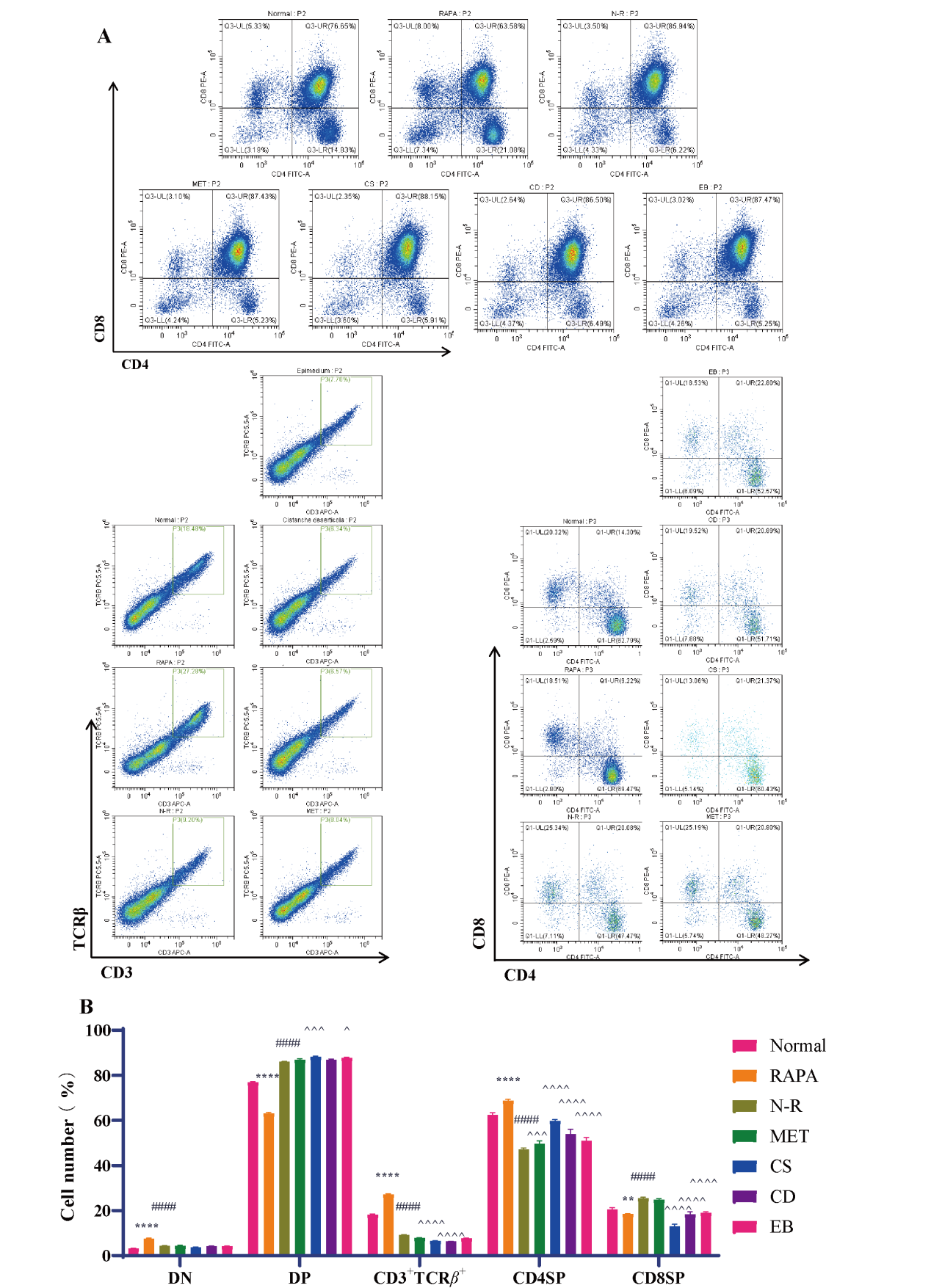
Figure 4 Kidney-tonifying Chinese medicine impacted the differentiation of thymic T cell subsets in mice. (A) Representative image of thymocyte differentiation in mouse thymus. CD4-CD8- (DN) cells differentiated into CD4+CD8+ DP cells and underwent positive selection. The percentage of DN cells (CD4-CD8-), DP cells (CD4+CD8+), and CD3+TCR β+ cells in the total cells. The percentage of mature single-positive CD8+ (CD8SP) cells (CD3+TCR β+CD4-CD8+) and mature single-positive CD4+(CD4SP) cells (CD3+TCR β+CD4+CD8-) to CD3+TCR β+ cells. (B) Representative images and statistical results of thymocyte differentiation in mouse thymus (n=3). Compared to the normal group, *p < 0.05, **p < 0.01, ***p < 0.001, ****p < 0.0001, compared to RAPA group, #p < 0.05, ##p < 0.01, ###p < 0.001, ####p < 0.0001, compared to N-R group, ^p < 0.05, ^^p < 0.01, ^^^p < 0.001, ^^^^p < 0.0001.
3.5 Kidney-tonifying Chinese medicine affected TREC level and the expression of thymosin and aging-related factors
TRECs are the mature biomarker for evaluating thymus output. We found that TRECs level was higher in the RAPA group than the normal group, but then explicitly waned after natural repair and thus suppressed thymus output. TREC downregulation was more apparent in the MET and TCM groups. The level of TRECs in the CD group was comparable to that observed in the normal group (Figure 5A).
We quantified expressions of aging-related genes and thymosin, revealing that the administration of RAPA resulted in upregulation of Sirt6, IL-1β, P21, IL-1α and TSLP mRNAs, as well as downregulation of Tα1 mRNA. Following natural repair, Sirt6, IL-1β, P21, IL-1α and TSLP levels were diminished, while Tα1 and PTMα mRNA levels were elevated. In contrast to N-R group, TCM groups exhibited reduced Sirt6, P21 and IL-1α levels. Meanwhile, CD apparently suppressed IL-1β and Tα1 mRNA expressions, while promoting PTMα mRNA expression. CS and EB repressed Sirt6, Tα1, PTMα and Tβ4 expressions (Figure 5B-I).
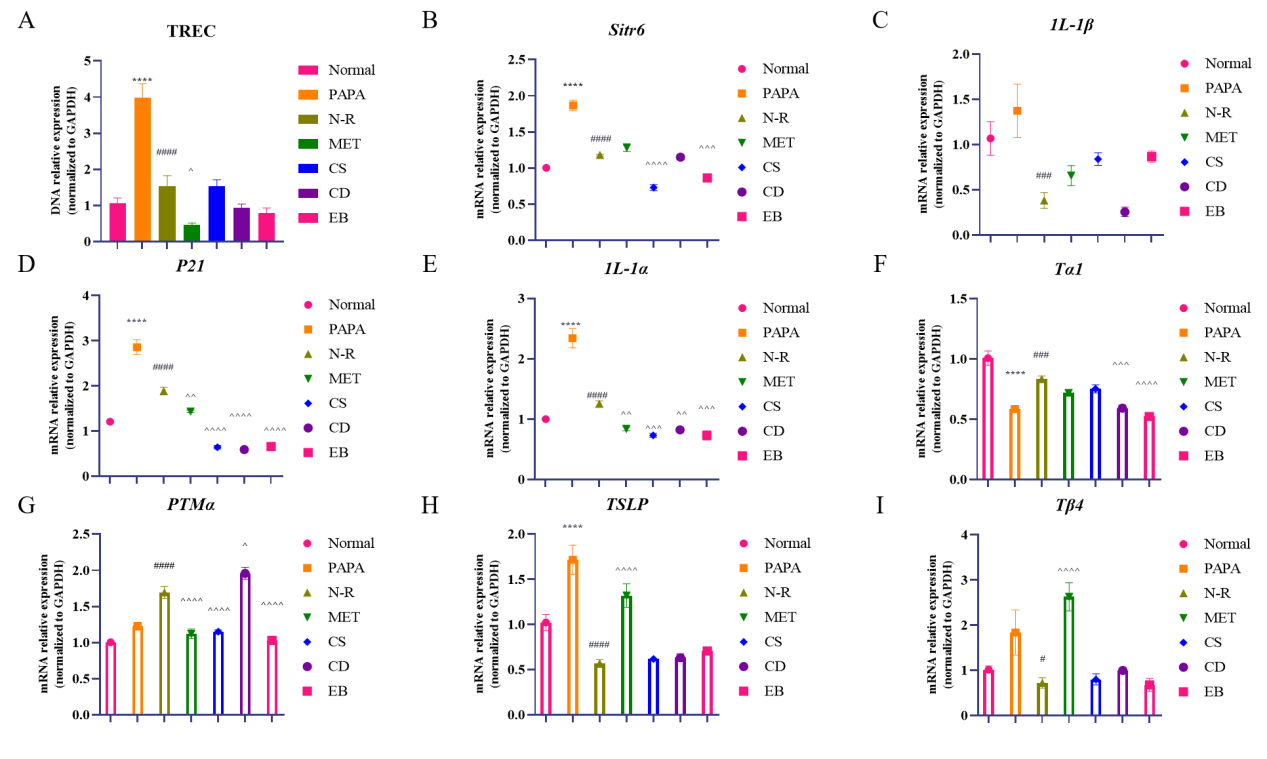
Figure 5 Kidney-tonifying Chinese medicine promoted thymus output and secretion functions in mice. (A) Relative level of TREC in spleen (n=6). (B) Sirt6 mRNA level (n=6). (C) IL-1β mRNA level (n=6). (D) P21 mRNA level (n=6). (E) IL-1α mRNA level (n=6). (F) Tα1 mRNA level (n=6). (G) PTMα mRNA level (n=6). (H) TSLP mRNA level (n=6). (I) Tβ4 mRNA level (n=6). Compared with the normal group, *p < 0.05, **p <0.01, ***p < 0.001, compared to RAPA group, #p < 0.05, ##p < 0.01, ###p < 0.001, compared to N-R group, ^p < 0.05, ^^p < 0.01, ^^^p < 0.001
4 Discussion
The development of the thymus begins in the embryonic period, and as it matures and reaches its functional peak, the thymus gradually degenerates, and leads to thymic atrophy, decreased T cells and function decline, ultimately causing a decline in the overall function of the immune system and accelerating the process of immunosenescence [3]. RAPA has been recognized as an anti-aging drug with immunomodulatory properties [10], and can regulate the activation and differentiation of T cells [11]. It has been documented that the combination of RAPA and MET can enhance anti-aging efficacy [13]. The waxing and waning of kidney qi mentioned in TCM is similar to the age-related changes in the thymus in modern medicine, and kidney deficiency has been confirmed to cause atrophy of immune organs, including the thymus [16], implying the close association between the kidneys and the production of immune cells. Kidney-tonifying Chinese medicines, such as CS, have multiple functions including scavenging free radicals, preventing oxidization and aging, resisting stress, regulating the immune and endocrine systems and improving sexual function [27]. CD has effects of enhancing human immunity, anti-aging, anti-inflammatory, and anti-fatigue [28]. EB can mediate immunity and promote the proliferation and development of osteoblasts [29].
The thymus shows pathological changes characterized by disordered cortex and medulla structures, particularly in the distribution and function of thymic epithelial cells, which are essential for thymic integrity and T cell development [30]. Our research revealed that short-term administration of RAPA significantly reduced the thymus size in mice, particularly the medulla area, which exhibited a marked decrease. The medulla developed a hollow cystic-like structure, and there was a reduction in CK5 expression. T cell subsets underwent significant changes. Correspondingly, mRNA levels were altered, with upregulation of Sirt6, IL-1β, P21, IL-1α, and TSLP mRNAs, while Tα1 mRNA was downregulated.
Following the discontinuation of the drug, the body weight of the mice increased along with an elevation in the thymus index and recovery of thymus structure. These effects were observed to be more pronounced in the TCM groups compared to the N-R group. Each TCM group showed differences in repair effect.
CS groups increased the proportion of DP thymocytes and promoted the differentiation of thymocytes into CD4SP. CD4SP is involved in the developmental pathway of medullary thymocytes and provides conditions for the development of thymocytes [31]. CS apparently suppressed SIRT6 mRNA expressions. SIRT6 is a longevity protein capable of inhibiting aging by facilitating DNA damage repair, maintaining the normal structure of chromosomes, and regulating energy metabolism as well as the senescence-associated secretory phenotype (SASP) [32].
While the CD group exhibited a significant increase in CK8 fluorescence intensity. At the same time, CD apparently suppressed P21, IL-1β and Tα1 mRNA expressions, while promoting PTMα mRNA expression. IL-1β is produced in excess, which leads to autoimmunity and causes autoinflammatory diseases [33]. P21 plays a crucial role in apoptosis and cell cycle regulation, the upregulation of which may suggest an immunosuppressive effect [34,35].
EB groups showed increased medulla with restored structure and more medullary epithelial cells. This group also demonstrated stronger CK5 fluorescence and clearer reticular nodes of mTECs. CD apparently suppressed IL-1α and Tβ4 mRNA expressions. IL-1α is a key senescence-associated proinflammatory cytokine that functions as a crucial upstream regulator of the SASP [36].
Kidney-tonifying Chinese medicines may maintain thymic cell numbers and TRECs levels via dampening the expressions of Sirt6, P21, IL-1α, and IL-1β, and hampered thymic cell apoptosis. Thymosin is a pivotal peptide hormone secreted by thymic stromal cells, primarily constituted by thymic epithelial cells. It mainly consists of two major families, α and β, and plays a vital regulatory role in the development, maturation, and proliferation of immune cells [37]. Changes in various peptide thymic hormones may reflect the disorder of the immune system, that is, the imbalance of thymosin levels may lead to a decline in immune function, which can be improved by CS, CD, and EB regulating the expressions of different thymosins. Gene expressions involve multiple regulatory steps, including activation, transcription, processing, degradation, and translation, ultimately forming functional proteins. This experiment studied the effects of CS, CD, and EB on the gene expressions of thymic epithelial cells, but their effects on protein levels remain to be further explored.
The mechanism of action was not further explored. However, the clinical significance is reflected in the effect of kidney tonifying traditional Chinese medicine on the regeneration of thymus, an important immune organ, which may have some clinical significance in anti-aging. A detailed exploration of the effects of these herbs on thymus regeneration may help to understand their mechanism of action and provide useful enlightenment for clinical application in the field of anti-aging. In addition, future studies can further explore the potential applications of these herbs in the regulation of immune system function and anti-aging therapy to expand their clinical significance and application scope.
This study demonstrated that kidney-tonifying herbs represented by CD can promote the regeneration of the thymus after RAPA treatment. The corresponding mechanism may be related to enhancing the thickness of the thymic capsule and cortex, as well as accelerating the reticular differentiation of medullary epithelial cells.
Back Matter
Acknowledgments
We appreciate the great experimental support from the Public Platform of Pharmaceutical Research Center, Academy of Chinese Medical Sciences, Zhejiang Chinese Medical University.
Conflicts of Interest
The author of this article, Jianli Gao, is a member of the editorial office of this journal. All procedures during the editorial review process were conducted strictly in accordance with the journal's policies, and the author was not involved in handling any part of the process.
Author Contributions
M.Z.: writing-original draft, conceptualization, methodology. X.W. and F.Z.: validation, data curation. S.H.: formal analysis, resources. Y.S. and Q.S.: writing-review and editing. J.G.: writing-review and editing, project administration, funding acquisition. All data were generated in-house, and no paper mill was used. All authors agree to be accountable for all aspects of work ensuring integrity and accuracy.
Ethics Approval and Consent to Participate
The animal study protocol was approved by the Ethics Committee of the Experimental Animal Research Center of Zhejiang Chinese Medicine University (Approval No. IACUC-20240311-12).
Funding
This work was supported by the Research Project of National Natural Science Foundation of China (82274621) and opening fund of the State Key Laboratory of Quality Research in Chinese Medicine, University of Macau (No. QRCM-OP21002).
Availability of Data and Materials
The analyzed data sets generated during the study are available from the corresponding author on reasonable request.
Supplementary Materials
The following supporting information can be downloaded at: https://ojs.exploverpub.com/index .php/jecacm/article/view/229/sup. Table S1: Wet weight, body weight and thymus index of mice; Table S2: List of primer sequences.
References
- Nishino M, Ashiku SK, Kocher ON, et al. The thymus: a comprehensive review. Radiographics 2006; 26(2): 335-348.
- Ohigashi I, Kozai M, Takahama Y. Development and developmental potential of cortical thymic epithelial cells. Immunological Reviews 2016; 271(1): 10-22.
- Aspinall R, Andrew D. Immunosenescence: potential causes and strategies for reversal. Biochemical Society Transactions 2000; 28(2): 250-254.
- Kanatli I, Akkaya B, Uysal H, et al. Analysis of TNF-related apoptosis-inducing ligand and receptors and implications in thymus biology and myasthenia gravis. Neuromuscul Disord 2017; 27(2): 128-135.
- Zdrojewicz Z, Pachura E, Pachura P. The Thymus: A Forgotten, But Very Important Organ. Advances In Clinical And Experimental Medicine 2016; 25(2): 369-375.
- Miller JF. The discovery of thymus function and of thymus-derived lymphocytes. Immunological Reviews 2002; 185: 7-14.
- Sciaky-Tamir Y, Hershkovitz R, Mazor M, et al. The use of imaging technology in the assessment of the fetal inflammatory response syndrome-imaging of the fetal thymus. Prenat Diagn 2015; 35(5): 413-419.
- Arriola Apelo SI, Lamming DW. Rapamycin: An InhibiTOR of Aging Emerges From the Soil of Easter Island. Journals of Gerontology Series A-biological Sciences and Medical Sciences 2016; 71(7): 841-849.
- Chi H. Regulation and function of mTOR signalling in T cell fate decisions. Nature Reviews Immunology 2012; 12(5): 325-338.
- Chen C, Liu Y, Liu Y, et al. mTOR regulation and therapeutic rejuvenation of aging hematopoietic stem cells. Sci Signal 2009; 2(98): ra75.
- Svatek RS, Ji N, de Leon E, et al. Rapamycin Prevents Surgery-Induced Immune Dysfunction in Patients with Bladder Cancer. Cancer Immunological Reviews 2019; 7(3): 466-475.
- Mannick JB, Lamming DW. Targeting the biology of aging with mTOR inhibitors. Nat Aging 2023; 3(6): 642-660.
- Strong R, Miller RA, Antebi A, et al. Longer lifespan in male mice treated with a weakly estrogenic agonist, an antioxidant, an α-glucosidase inhibitor or a Nrf2-inducer. Aging Cell 2016; 15(5): 872-884.
- Roberts MN, Wallace MA, Tomilov AA, et al. A Ketogenic Diet Extends Longevity and Healthspan in Adult Mice. Cell Metabolism 2017; 26(3): 539-546.e5.
- Li ZD, Chen YX, Gong BY, et al. Preliminary study on delaying aging induced thymus degeneration in SAMP6 mice with Bazi Bushen capsule. Chinese Pharmacological Bulletin 2024; 40(6): 1186-1192.
- Li Z, Liu R, Kang X, et al. Study on establishment of kidney deficient aging model and comparison with D-galactose induced aging model. Zhongguo Zhong Yao Za Zhi 2012; 37(16): 2435-2438.
- Liu HP, Chang RF, Wu YS, et al. The Yang-Tonifying Herbal Medicine Cynomorium songaricum Extends Lifespan and Delays Aging in Drosophila. Evidence-Based Complementary and Alternative Medicine 2012; 2012: 735481.
- Li TM, Huang HC, Su CM, et al. Cistanche deserticola extract increases bone formation in osteoblasts. Journal of Pharmacy and Pharmacology 2012; 64(6): 897-907.
- An R, Li B, You LS, et al. Improvement of kidney yang syndrome by icariin through regulating hypothalamus-pituitary-adrenal axis. Chinese Journal of Integrative Medicine 2015; 21(10): 765-771.
- Kang L, Zhang Y, Zhou L, et al. Structural Characterization and Discrimination of Morinda officinalis and Processing Morinda officinalis Based on Metabolite Profiling Analysis. Frontiers in Chemistry 2022; 9: 803550.
- Song S, Gao Z, Lei X, et al. Total Flavonoids of Drynariae Rhizoma Prevent Bone Loss Induced by Hindlimb Unloading in Rats. Molecules 2017; 22(7): 1033.
- State Pharmacopoeia Commission. Chinese Pharmacopoeia; China Pharmaceutical Science and Technology Press: Beijing, China, 2020; 340-341.
- Rao X, Huang X, Zhou Z, et al. An improvement of the 2ˆ(-delta delta CT) method for quantitative real-time polymerase chain reaction data analysis. Biostat Bioinforma Biomath 2013; 3(3): 71-85.
- Pan XH, Lin QK, Yao X, et al. Umbilical cord mesenchymal stem cells protect thymus structure and function in aged C57 mice by downregulating aging-related genes and upregulating autophagy- and anti-oxidative stress-related genes. Aging (Albany NY) 2020; 12(17): 16899-16920.
- Heinonen KM, Vanegas JR, Brochu S, et al. Wnt4 regulates thymic cellularity through the expansion of thymic epithelial cells and early thymic progenitors. Blood 2011; 118(19): 5163-5173.
- Love PE, Bhandoola A. Signal integration and crosstalk during thymocyte migration and emigration. Nature Reviews Immunology 2011; 11(7): 469-477.
- Miao M, Yan X, Guo L, et al. Effect of Cynomorium total flavone on depression model of perimenopausal rat. Saudi Journal of Biological Sciences 2017; 24(1): 139-148.
- Tian S, Miao M, Bai M, et al. Phenylethanoid Glycosides of Cistanche on menopausal syndrome model in mice. Saudi Pharmaceutical Journal 2017; 25(4): 537-547.
- Wang Z, Wang D, Yang D, et al. The effect of icariin on bone metabolism and its potential clinical application. Osteoporos International 2018; 29(3): 535-544.
- akada K, Ohigashi I, Kasai M, et al. Development and function of cortical thymic epithelial cells. Current Topics in Microbiology and Immunology 2014; 373: 1-17.
- Li J, Li Y, Yao JY, et al. Developmental pathway of CD4+CD8- medullary thymocytes during mouse ontogeny and its defect in Aire-/- mice. Proceedings of the national academy of sciences of the united states of america 2007; 104(46): 18175-80.
- Li Y, Jin J, Wang Y. SIRT6 Widely Regulates Aging, Immunity, and Cancer. Frontiers in Oncology 2022; 12: 861334.
- Bent R, Moll L, Grabbe S, et al. Interleukin-1 Beta-A Friend or Foe in Malignancies? International Journal of Molecular Sciences 2018; 19(8): 2155.
- Karimian A, Ahmadi Y, Yousefi B. Multiple functions of p21 in cell cycle, apoptosis and transcriptional regulation after DNA damage. DNA Repair (Amst) 2016; 42: 63-71.
- Shamloo B, Usluer S. p21 in Cancer Research. Cancers (Basel) 2019; 11(8): 1178.
- McCarthy DA, Clark RR, Bartling TR, et al. Redox control of the senescence regulator interleukin-1α and the secretory phenotype. Journal Of Biological Chemistry 2013 Nov 8;288(45):32149-32159.
- Shi D, Shui Y, Xu X, et al. Thymic function affects breast cancer development and metastasis by regulating expression of thymus secretions PTMα and Tβ15b1. Translational Oncology 2021; 14(1): 100980.